Hybrid closed loop system now available for people with type 1 diabetes
Latest device follows draft guidance published by National Institute for Health and Care Excellence (NICE) earlier this year.
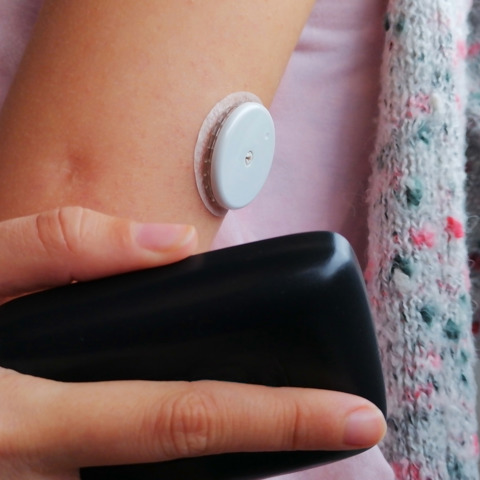
The first continuous glucose monitor (CGM) is available now that can offer readings every minute for people living with type 1 diabetes.
The FreeStyle Libre 3 sensor from Abbott is now authorised to work with Ypsomed’s mylife YpsoPump and CamDiab's CamAPS FX mobile app offering a new form of insulin delivery to help people living with type 1 diabetes with their self-management of the condition.
These technologies work together to form an intelligent process to deliver insulin based on real-time glucose readings via an automated insulin delivery system (AID), designed to help people living with type 1 diabetes improve how they manage their condition daily.
A hybrid closed loop system works by continuously monitoring a person’s glucose levels based on real-time readings from the glucose sensor with an algorithm in the app to calculate and deliver the right amount of insulin as needed.
This removes the guesswork of insulin dosing and can improve health outcomes by helping those living with Type 1 diabetes to improve their quality of life and reach better treatment targets.

In January draft guidance was published by NICE that recommended the use of hybrid closed loop systems, such as the one announced today by Abbott, for people with type 1 diabetes that require pump therapy.
Recent NHS trials of the technology in use provided figures which allowed NICE to consider wider use of the technology which allows a person with type 1 diabetes to go about their day-to-day life without having to monitor if their blood glucose levels are too high or too low.
If type 1 diabetes is not well controlled, people are at risk of long-term complications of hyperglycaemia (high blood glucose levels), including blindness, amputations, or kidney problems.
The hybrid closed loop system is made up of a continuous glucose monitor (CGM) sensor attached to the body. This sends information to an insulin pump, worn on the body. This uses the figures to run a mathematical calculation and work out how much insulin needs to be delivered into the body to keep blood glucose levels within a healthy range.
The new glucose sensing technology are the smallest, thinnest and most discreet sensors and last for up to 14 days and are now used by more than 4.5 million people across more than 60 countries, by providing breakthrough technology that is accessible and affordable.
Professor Partha Kar, OBE, national specialty advisor, diabetes at NHS England said: “It is wonderful to see the coming together of Ypsomed’s mylife YpsoPump; CamAPX FX algorithm and Abbott's FreeStyle Libre 3 sensor into one hybrid closed loop system – creating exciting opportunities for people living with type 1 diabetes. This type of advanced technology has the potential to change people’s lives for the better, so I’m pleased to see companies continue to innovate and bring functionality like this together, to allow for broader choice for patients and healthcare professionals.”
I would like to make a regular donation of
There are lots of ways to raise money to support
people living with all forms of diabetes.
Bake, Swim, Cycle, Fly ... Do It For DRWF!
Fundraise with us
Recent News


